Technical Article:
PFAS Atmospheric Deposition & Transport Modeling
Cross-Media Transport Integration
Executive Overview
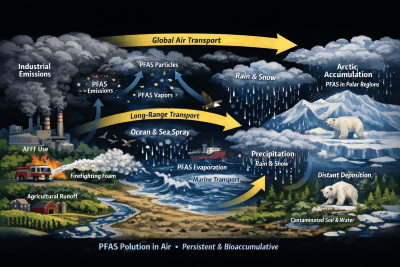
Per- and polyfluoroalkyl substances (PFAS) are often associated with groundwater contamination and drinking water impacts. However, atmospheric transport and deposition are becoming more important environmental loading pathways. PFAS released into the atmosphere through industrial emissions, stacks, fugitive emissions, thermal processes, waste water treatment processes, AFFF-related volatilization/aerosols, and resuspended dust, can undergo long-range transport, chemical transformation, and subsequently deposit downwind through dry deposition (particles/gases contacting surfaces) and wet deposition (scavenging by rain/snow).
Once deposited, PFAS can migrate across media—soil → vadose zone → groundwater, soil → runoff → surface water, and surface water → sediment/biota—creating “cross-media” exposure pathways that are often underestimated when air, soil, and groundwater are evaluated separately. Understanding these processes requires application of advanced atmospheric dispersion and chemical transport models such as AERMOD, CMAQ, CAMx, and SCICHEM. This article summarizes common air deposition modeling equation frameworks and shows how to integrate deposition outputs into soil/vadose/groundwater multimedia transport.
AA GeoEnvironmental provides independent technical review of PFAS atmospheric transport and deposition modeling, evaluating model assumptions, uncertainty, and cross-media linkage to vadose zone, groundwater, and watershed contamination.
Atmospheric Transport Processes
Atmospheric transport of PFAS occurs through several fundamental physical and chemical processes. These mechanisms determine how PFAS emissions move through the atmosphere and where they ultimately deposit in the environment.
Atmospheric Advection
PFAS compounds released into the atmosphere are transported downwind by regional air masses. The distance of transport depends on:
- wind speed
- atmospheric stability
- mixing height
- PFAS compounds volatility.
Some PFAS precursor compounds can travel hundreds or thousands of kilometers before chemical transformation or deposition occurs.
Turbulent Dispersion
Atmospheric turbulence spreads PFAS plumes both vertically and horizontally. Turbulence is influenced by:
- solar heating
- terrain roughness
- atmospheric stability conditions
Dispersion causes contaminant plumes to dilute as they move away from the source.
Chemical Transformation
Some PFAS compounds released into the atmosphere exist as volatile precursor compounds. These precursors can undergo atmospheric oxidation reactions that produce persistent perfluoroalkyl acids such as PFOA, PFNA, and PFHxA.
This process allows relatively short-lived atmospheric compounds to form extremely persistent PFAS that deposit into ecosystems.
Long-Range Atmospheric Transport
PFAS compounds exhibit chemical stability, allowing them to persist in the atmosphere and be transported over regional and global distances. Long-range transport explains detection of PFAS in remote environments including:
- Arctic ecosystems
- high-altitude watersheds
- remote precipitation
These findings highlight the importance of atmospheric modeling when evaluating PFAS environmental distribution.
Deposition Processes
Wet Deposition
Wet deposition removes PFAS chemicals from the atmosphere by precipitation such as rainfall, snowfall, fog, and cloud scavenging. Wet deposition is often the dominant PFAS atmospheric removal mechanism.
Dry Deposition
Dry deposition occurs when airborne contaminants settle onto surfaces without precipitation. This process includes particles gravitational settling, surface absorption, and turbulent transfer.
PFAS Air Modeling Frameworks
Near-Field Dispersion Models
AERMOD: This model simulate atmospheric dispersion from individual emission sources over relatively short distances, steady-state plume assumption, and dry deposition estimates.
Puff Models
CALPUFF: Lagrangian puff dispersion, long-range transport complex terrain, handles non-steady meteorology, and limited chemical representation.
SCICHEM: Reactive puff models simulate the evolution of contaminant plumes as they move through changing meteorological conditions.
Regional Chemical Transport Models
CMAQ and CAMx: Eulerian regional models simulate atmospheric chemistry and transport across large geographic domains. These models account for: emissions from multiple sources, wet and dry deposition. Regional models are often required when evaluating watershed PFAS loading from atmospheric deposition.
Cross-Media Deposition & Transport
Atmospheric PFAS deposition represents a cross-media contamination pathway linking air emissions to groundwater and ecosystem impacts.
The general pathway includes the following stages:
Atmospheric Deposition
PFAS compounds deposit onto land surfaces through wet and dry deposition.
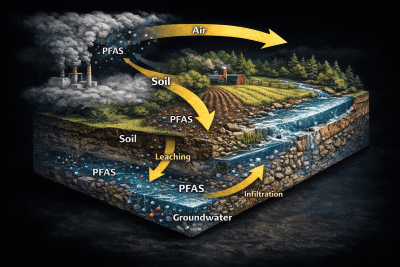
Soil Retention
Deposited PFAS may bind to soil particles or organic matter. PFAS interact with the air-water interface in the vadose zone, affecting vertical migration.
Surface Water Transport
PFAS deposited onto land surfaces may be transported into streams and lakes through surface runoff, stormwater transport, and erosion.
Groundwater Recharge
A portion of deposited PFAS may infiltrate through the soil and vadose zone, eventually reaching groundwater. This pathway is particularly relevant for drinking water aquifers and watershed recharge areas.
Core mathematical Equations
Atmospheric advection–diffusion–reaction (Eulerian) equation
Governing equation (3-D continuity for a species C):
$$
\frac{\partial C}{\partial t}
=
– \nabla \cdot (\mathbf{u} C)
+ \nabla \cdot (K \nabla C)
+ E
– (L_{\text{dry}} + L_{\text{wet}})
+ R_{\text{chem}}
$$
Definitions
- C = air concentration of a PFAS species
- t = time
- u = wind (advection) vector
- K = turbulent (eddy) diffusivity tensor
- E = emissions source term
- Ldry = dry deposition loss rate
- Lwet = wet deposition loss rate
- Rchem = net chemical production/loss (e.g., precursor oxidation → PFCA formation)
This is the backbone of chemical transport models (CAMx/CMAQ/WRF-Chem/GEOS-Chem) and many dispersion models that indicates: winds move PFAS, turbulence spreads it, emissions add it, deposition removes it, and chemistry transforms it.
AERMOD
Steady state continuous point-source Gaussian plume dispersion:
$$
C(x,y,z)=\frac{Q}{2\pi U \sigma_y \sigma_z}
\exp\!\left(-\frac{y^2}{2\sigma_y^2}\right)
\left[
\exp\!\left(-\frac{(z-H)^2}{2\sigma_z^2}\right)
+
\exp\!\left(-\frac{(z+H)^2}{2\sigma_z^2}\right)
\right]
$$
- C(x,y,z) = concentration at receptor
- Q = emission rate
- U = mean wind speed at plume height
- σy,σz = lateral/vertical dispersion parameters (m) (depend on stability, turbulence, distance)
- H = effective stack height = physical height + plume rise
- x = downwind distance (m), y = crosswind offset, z = height above ground
- The z+Hz+Hz+H term represents “reflection” at the ground boundary
The Gaussian plume provides the mathematical foundation for several near-field dispersion models. AERMOD is fundamentally an advanced Gaussian model. It is not a chemistry model and is generally not a defensible tool for regional PFAS deposition attribution.
Time-Varying Plumes: Gaussian Puff
CALPUFF
Lagrangian puff dispersion model designed to simulate transport under time-varying meteorological conditions.
Puff Concentration Equation:
$$
C(x,y,z,t)=\frac{M(t)}{(2\pi)^{3/2}\sigma_x\sigma_y\sigma_z}
\exp\!\left(
-\frac{x^2}{2\sigma_x^2}
-\frac{y^2}{2\sigma_y^2}
-\frac{z^2}{2\sigma_z^2}
\right)
$$
Puff mass balance (losses + chemistry)
$$
\frac{dM}{dt}
=
-(\lambda_{\text{dry}}+\lambda_{\text{wet}})M
+
P_{\text{chem}}
–
L_{\text{chem}}
$$
- M(t) = mass in puff
- σx,σy,σz = puff spread parameters
- λdry, λwet = first-order removal rates
- Pchem, Lchem = chemical production/loss terms
CALPUFF simulates contaminant transport using discrete puffs that move with the wind field and expand due to turbulence.
Gaussian puff models handle changing winds and time-varying emissions better than steady-state plumes.
SCICHEM
Reactive Lagrangian puff model capable of simulating atmospheric chemistry.
Puff Transport Equation:
$$
\frac{dx}{dt} = u(x,t)
$$
$$
\frac{\partial C}{\partial t}
+
\mathbf{U} \cdot \nabla C
=
\nabla \cdot (K \nabla C)
+
R(C)
$$
- x = puff position vector
- u(x,t) = wind velocity field
- K = turbulent diffusivity tensor
- R(C) = chemical production–loss term (nonlinear reaction kinetics)
Chemical Reaction Kinetics:
$$
\frac{dC_i}{dt} = P_i – L_i
$$
- Ci = concentration of chemical species i
- Pi = production rate of species i
- Li = loss rate of species i
SCICHEM is particularly relevant where PFAS precursor transformation materially influences deposition patterns.
CMAQ
CMAQ is a three-dimensional Eulerian chemical transport model used for regional air quality simulations.
Governing Atmospheric Transport Equation:
$$
\frac{\partial C}{\partial t}
=
– \nabla \cdot (\mathbf{u}C)
+ \nabla \cdot (K \nabla C)
+ E
– D
+ R
$$
CAMx
CAMx employs a similar Eulerian framework, however, incorporating supplementary modules for enhanced chemistry and source allocation.
Eulerian Transport Equation:
$$
\frac{\partial C}{\partial t}
=
-\left(
u \frac{\partial C}{\partial x}
+
v \frac{\partial C}{\partial y}
+
w \frac{\partial C}{\partial z}
\right)
+
\nabla \cdot (K \nabla C)
+
E
–
D
+
R
$$
u, v, w = wind velocity components in x, y, z directions. Other variables defined as above.
CAMx explicitly represents three-dimensional wind transport and atmospheric chemistry interactions. The model is widely used for regional air quality and deposition studies.
PFAS Atmospheric Deposition Fluxes
Dry Deposition Flux
Flux and Velocity:
$$
F_{\text{dry}} = v_d C
$$
Deposition velocity resistances:
$$
v_d = \frac{1}{r_a + r_b + r_c}
$$
- Fdry = dry deposition flux
- vd = deposition velocity
- C = near-surface air concentration
- ra = aerodynamic resistance
- rb = quasi-laminar boundary resistance
- rc = surface/canopy resistance
Dry deposition often drives near-source soil loading. For PFAS, Vd can be highly uncertain as it depends on species, land cover, meteorology, and physicochemical properties.
Wet Deposition Flux
Flux
$$
F_{\text{wet}} = P\,C_{\text{rain}}
$$
First-order scavenging representation (in & below cloud)
$$
\frac{dC}{dt} = -\Lambda C \;\Rightarrow\; C(t) = C_0 e^{-\Lambda t}
$$
- Fwet = wet deposition flux
- P = precipitation rate
- Crain = PFAS concentration in precipitation
- Λ = scavenging coefficient
- C0 = initial air concentration
Wet deposition is the main regional removal mechanism for many PFAS species, and precipitation data are useful since they directly affect watershed loading rates.
PFAS Precursors Atmospheric Transformation
Many PFAS emissions include volatile precursor, e.g., fluorotelomer alcohols. First-order reaction kinetics may produce persistent perfluoroalkyl acids from atmospheric precursor oxidation.
First-order precursor loss:
$$
\frac{dC_p}{dt} = -k C_p \;\Rightarrow\; C_p(t) = C_{p,0} e^{-kt}
$$
Product (PFCA) formation with yield, Y:
$$
\frac{dC_a}{dt} = YkC_p – k_a C_a
$$
- Cp = precursor concentration in air
- Ca = acid (e.g., PFCA) concentration in air
- k = precursor reaction rate constant
- Y = molar or mass yield fraction
- ka = loss rate for acid species
This mechanism explains how persistent PFAS acids may form during atmospheric transport. Even if acids are not emitted strongly, deposition can still be high if precursors are emitted and transform efficiently.
Gas - Particle Partitioning
Generic phase partition fraction:
$$
f_p = \frac{C_p}{C_p + C_g}
$$
- Cp = particle-phase concentration
- Cg = gas-phase concentration
- fp = particle fraction
Particle-phase PFAS typically deposit faster than gas-phase PFAS. If phase partitioning is incorrect, deposition patterns may also be incorrect, even when transport is accurate.
Cross-Media PFAS Transport & Deposition
Air–Water Exchange
Two-film style net flux:
$$
J_{aw} = k_{aw}\left(C_w – H’ C_a\right)
$$
- Jaw = net air-to-water flux
- kaw = overall mass transfer coefficient
- Cw = dissolved concentration in surface water
- Ca = air concentration
- H′ = dimensionless Henry’s constant
This establishes a direct connection between air and water. It’s most important for precursors that are volatile/neutral.
Watershed & Groundwater Recharge
Annual areal load from deposition:
$$
L_{\text{annual}} = \int_{0}^{T} \left(F_{\text{dry}} + F_{\text{wet}}\right)\, dt
$$
Groundwater Recharge
$$
C_{\text{recharge}}=\frac{f_{\text{inf}}\,L_{\text{annual}}}{R}
$$
- L_annual = annual areal load
- T = integration time
- finf = fraction of deposited mass that infiltrates to recharge
- R = recharge depth per year
- Crecharge = estimated recharge concentration
This represents the critical connection between air and groundwater. It should be integrated with soil and vadose zone processes to ensure sound site decisions.
Uncertainty in PFAS Atmospheric Modeling
Major sources of uncertainty in PFAS Atmospheric Modeling are emission rates, reaction mechanisms, meteorological variability, and deposition parameters.
Monte Carlo simulation can be used to evaluate the uncertainty of the listed parameters.
$$
Y_i = f(p_{1i}, p_{2i}, \ldots, p_{ni})
$$
Sample mean
$$
\bar{Y} = \frac{1}{N}\sum_{i=1}^{N} Y_i
$$
Variance
$$
\sigma_Y^2 = \frac{1}{N-1} \sum_{i=1}^{N} (Y_i – \bar{Y})^2
$$
Confidence interval
$$
Y_{\text{upper/lower}} = \bar{Y} \pm z \sigma_Y
$$
- Yi = output for run i
- pji = sampled parameter value for parameter j in run i
- N = number of simulations
- Yˉ = mean output
- σY = standard deviation of outputs
- z = z-score (e.g., 1.96 for ~95% CI)
Monte Carlo can produce probabilistic ranges of model outcomes, instead of a single deterministic outcome.
References
Armitage, J. M., Schenker, U., Scheringer, M., et al. (2009). Modeling the global fate and transport of perfluorooctanoic acid (PFOA). Environmental Science & Technology, 43(4), 1134–1140.
Byun D., Schere K. L. (2006). Review of the Governing Equations, Computational Algorithms, and Other Components of the Models-3 Community Multiscale Air Quality (CMAQ) Modeling System. Applied Mechanics Reviews, 59(2), 51–77.
Holmes, N. S., & Morawska, L. (2006). A review of dispersion modeling and its application to atmospheric pollution. Atmospheric Environment, 40(30), 5902–5928.
Scire, J. S., Strimaitis, D. G., & Yamartino, R. J. (2000). CALPUFF dispersion modeling system user guide.
U.S. Environmental Protection Agency. (2023). AERMOD modeling system user guide. U.S. EPA
Wallington, T. J., Hurley, M. D., Xia, J., et al. (2006). Formation of C7F15COOH (PFOA) and Other Perfluorocarboxylic Acids during the Atmospheric Oxidation of 8:2 Fluorotelomer Alcohol
PFAS Atmospheric Modeling FAQs
PFAS atmospheric transport and deposition modeling simulates how per- and polyfluoroalkyl chemicals travel through the atmosphere, undergo chemical transformations, and deposit on environmental surfaces. These models use meteorological data, emissions inventories, and chemical reaction mechanisms to predict PFAS concentrations in the atmosphere and their deposition in soils, surface water, and groundwater recharge areas.
Recent research suggests that atmospheric transport may spread PFAS contamination across broad areas. Many PFAS compounds or precursor chemicals can be released into the atmosphere and travel long distances before depositing on land or water. Thus, environmental investigators increasingly evaluate atmospheric transport pathways when determining potential sources of PFAS contamination.
Some PFAS behave differently in the atmosphere. Volatile precursor molecules like fluorotelomer alcohols are more likely to move far in the atmosphere. Airborne chemical processes can convert precursor molecules into persistent PFAS acids like PFOA or PFNA. Many ionic PFAS acids are less volatile and tend to deposit near emission sources.
Meteorological conditions significantly affect PFAS transport in the atmosphere. Wind direction dictates the path of contaminant plumes, whereas the wind speed affects how far they travel. Atmospheric turbulence and mixing height influence the vertical and horizontal dispersion of pollutants. Precipitation events can rapidly remove PFAS from the atmosphere via wet deposition.
PFAS Atmospheric modeling can help identify potential contamination sources. Investigators can determine if atmospheric emissions from certain facilities cause contamination by comparing modeled deposition patterns to environmental contamination data. However, modeling results should always be interpreted with monitoring and site investigation findings.
Several factors contribute to the uncertainty inherent in PFAS atmospheric modeling. Primary sources include emission rates, incomplete emission inventories, atmospheric reaction mechanisms, limited PFAS deposition measurements, and meteorological variability. Sensitivity and uncertainty analyses are often used to evaluate the impact of these uncertainties on modeling results.
Investigation of PFAS atmospheric transport is essential when there are potential emission sources, unknown contamination sources, suspected regional PFAS deposition, and the potential presence of precursor compounds in emissions. In certain instances, atmospheric deposition can serve as an important pathway leading to environmental contamination.
PFAS Atmospheric modeling often impacts environmental investigations, remediation approaches, and decision-making processes. An independent technical review identifies modeling limitations that may affect environmental conclusions and ensures that modeling assumptions, parameter values, and interpretations are scientifically valid.
