PFAS Modeling FAQs
Explore PFAS vadose zone modeling FAQs, including PFAS leaching to groundwater, air-water interfacial adsorption, precursor transformation, calibration, sensitivity, uncertainty analysis.
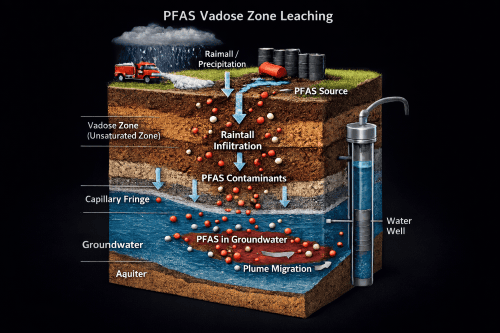
PFAS vadose zone fate and transport modeling evaluates how PFAS moves through unsaturated soil above the water table and whether it will remain stored in soil or leach downward to groundwater over time.
PFAS transport is different because many PFAS compounds strongly adsorb at the air-water interface in unsaturated soils, which can significantly retard transport and create long-term contaminant storage in the vadose zone.
A defensible PFAS vadose zone model may include variably saturated flow, advection, dispersion, solid-phase adsorption, air-water interfacial adsorption, rate-limited desorption, precursor transformation, and preferential flow where site conditions justify those processes.
Preferential flow should be considered when a site has fractured media, structured soils, perched conditions, rapid infiltration behavior, or field evidence of early breakthrough that matrix flow alone cannot explain.
Rate-limited desorption is important because it helps explain why PFAS can continue to leach long after the main source is gone and why contamination may decline more slowly than predicted by simple equilibrium models.
PFAS precursor transformation is the conversion of precursor compounds into terminal PFAS in the subsurface. It can sustain or increase long-term PFAS loading to groundwater even when measured terminal PFAS concentrations appear modest at the start of an investigation. Precursor transformation modeling is most appropriate when site history, chemistry, or analytical evidence indicates that precursors are present and may materially affect future PFAS loading.
PFAS precursor transformation is usually most defensibly represented as a lumped reactive-transport approximation of net precursor loss and daughter-PFAS generation rather than a universal mechanistic reaction. This captures the net effect of precursor degradation and daughter-PFAS formation without resolving every intermediate compound or pathway.
A weighted least-squares objective function is a mathematical way of quantifying mismatch between observed and simulated values while giving more influence to more reliable or relevant data. While, regularization is a calibration approach that helps keep parameter estimates within realistic ranges when the model contains poorly constrained parameters such as air-water interfacial terms or kinetic mass-transfer coefficients.
Sensitivity analysis evaluates which model parameters most influence predictions such as time to groundwater arrival, peak concentration, retained vadose zone mass, or long-term mass discharge. It helps identify which assumptions matter most and which field measurements would most improve model confidence. Uncertainty analysis estimates the plausible range of model outcomes after considering uncertainty in parameters, observations, assumptions, future scenarios, and model structure. It is important because a single best-fit model can hide major uncertainty in long-term PFAS leaching, plume persistence, and groundwater impact.
Red flags include, but are not limited to, neglecting air-water interfacial adsorption, using generic PFAS parameters without justification, relying on one-dimensional assumptions for clearly heterogeneous sites, disregarding sensitivity and uncertainty analysis, and overstating precursor transformation certainty.
Common limitations include uncertain interfacial area estimates, limited precursor data, poorly constrained kinetic parameters, transferability of lab parameters to field scale, and incomplete representation of heterogeneity or preferential flow.
Vadose zone modeling can estimate whether continued leaching from unsaturated soils is likely to sustain groundwater concentrations even if the saturated plume appears stable or partially controlled. PFAS vadose zone modeling can help estimate long-term source loading, evaluate source-zone remedies, compare remedial scenarios, assess whether monitored retention is plausible, and improve long-term groundwater protection strategies.
Screening-level models use simplified assumptions and fewer parameters, while advanced models may include transient unsaturated flow, interfacial adsorption, kinetic desorption, precursor transformation, and preferential flow. Advanced modeling is often warranted for large or complex sites, litigation support, remedial design, high-stakes risk decisions, heterogeneous geology, precursor-rich sources, or situations where long-term groundwater loading is a key concern.
AA GeoEnvironmental provides independent technical review of PFAS conceptual site models, governing equation selection, parameter defensibility, calibration strategy, sensitivity analysis, uncertainty evaluation, precursor transformation interpretation, and long-term leaching to groundwater.